Summary
Fibromyalgia is a chronic disorder marked by widespread musculoskeletal pain and increased sensitivity to stimuli. The acid-sensing ion channel 3 (ASIC3) is believed to play a key role in its pathogenesis, making it a potential therapeutic target. Artemisinin, a natural compound from Artemisia annua, has shown promise as a pain modulator. This study examined Artemisinin’s effect on ASIC3 using both in vivo and in silico approaches in a rat model of fibromyalgia. Fibromyalgia-like symptoms were induced in rats, and pain behaviors were assessed. Artemisinin was then administered to evaluate its impact on pain sensitivity. Results showed a significant reduction in pain responses following reduction in pain responses following Artemisinin treatment. In parallel, molecular docking studies revealed that Artemisinin binds to specific regions of ASIC3, potentially affecting its function. Furthermore, analysis suggests that Artemisinin may downregulate ASIC3 expression, providing a potential mechanism for its analgesic effect. These findings indicate that Artemisinin modulates ASIC3 activity and reduces pain sensitivity in fibromyalgia, offering a promising therapeutic avenue. Both behavioral and molecular data support this interaction, highlighting the potential of Artemisinin in fibromyalgia management. Further studies are needed to fully understand its mechanism and to validate its clinical applicability in human fibromyalgia treatment.
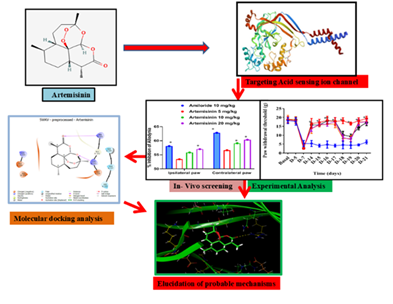
Graphical Abstract
Keywords
Fibromyalgia; ASIC3; Artemisinin; Pain modulation; Molecular docking.
Introduction
Fibromyalgia syndrome (FMS) is a complex chronic pain disorder affecting approximately 2-4% of the global population, with higher prevalence in women [1]. It is characterized by widespread musculoskeletal pain, accompanied by fatigue, sleep disturbances, and cognitive impairments [2]. Despite extensive research, the pathophysiological mechanisms underlying fibromyalgia remain incompletely understood, presenting substantial challenges for developing effective characterized by widespread.
Increasing evidence suggests that peripheral and central sensitization of nociceptive pathways plays a crucial role in fibromyalgia pathogenesis [3]. Acid-sensing ion channels (ASICs), particularly ASIC3, have emerged as key contributors to pain signaling in various chronic pain conditions [4]. ASIC3 is abundantly expressed in peripheral sensory neurons, especially in dorsal root ganglia (DRG), and is activated by extracellular acidosis, which commonly occurs during tissue inflammation and injury [5]. Several studies have reported elevated ASIC3 expression in animal models of chronic pain, including fibromyalgia [6]. ASIC3, predominantly expressed in skeletal muscle afferents, plays a key role in chronic pain models. In rodent models mimicking FM, such as the repeated intramuscular acid injection or reserpine model, ASIC3 expression and activity are significantly upregulated. Pharmacological blockade of ASIC3 (e.g., with APETx2) prevents development of widespread pain, and ASIC3 knockout mice resist hyperalgesia—highlighting ASIC3’s critical role in peripheral nociceptive sensitization in FM.
Current pharmacological treatments for fibromyalgia provide limited relief and are often associated with adverse effects [7]. Therefore, there is an urgent need to develop novel therapeutic approaches with improved efficacy and safety profiles. Natural compounds represent promising candidates for drug development due to their structural diversity and potentially favorable safety profiles [8].
Artemisinin, a sesquiterpene lactone isolated from the herb Artemisia annua (sweet wormwood) containing a unique peroxide bridge, which is crucial for its biological activity. It is well-known for its antimalarial properties [9]. Beyond its antiparasitic effects, artemisinin and its derivatives have demonstrated anti-inflammatory and analgesic properties in various experimental models [10-13]. Recent studies demonstrate its ability to modulate key inflammatory pathways—such as NF‑κB, MAPK, PI3K/Akt, and TGF‑β—which contribute to fibrosis, joint inflammation, and neuropathic pain [10-11]. In preclinical pain models, artemisinin and derivatives (e.g. artesunate) have yielded analgesic effects in neuropathic and inflammatory contexts, attenuating pain behaviors and reducing pro‑inflammatory mediator levels suggesting potential relevance to FM [11].
Despite its multifaceted biological activities, the direct impact of artemisinin on ASIC3 has not yet been established. Given that ASIC3-mediated signaling underpins the development of widespread hyperalgesia in FM, and artemisinin modulates pain‑related ion channels and inflammation, it is rational to explore whether artemisinin can modulate ASIC3 function, thereby alleviating FM‑like pain. However, the potential modulatory effects of artemisinin on ASIC3 and its therapeutic implications for fibromyalgia have not been explored. The present study aimed to investigate the modulatory role of artemisinin on ASIC3 in a rat model of fibromyalgia using both in vivo and in silico approaches. We propose that in a rat model of fibromyalgia, systemic administration of artemisinin will attenuate ASIC3-mediated hyperalgesia via direct or indirect modulation of ASIC3, resulting in reduced pain behaviors. We further hypothesize that in silico docking will support a direct interaction between artemisinin and ASIC3, elucidating the mechanistic underpinnings of its analgesic effect.
Results
In vivo studies
General observations: The current study aimed to evaluate the effects of Artemisinin on mechanical allodynia induced by repeated intramuscular injections of acidic saline. Twenty-four hours after the second injection, the mechanical withdrawal threshold of the ipsilateral paw decreased from 18.44 ± 0.2 g to 6.013 ± 0.71 g (n = 30), as summarized in Figure 1. Similarly, the contralateral paw showed a reduction from 19.41 ± 0.87 g to 7.34 ± 0.51 g. This significant increase in sensitivity persisted for three weeks after the second unilateral injection for both paws. No significant difference was observed between the withdrawal thresholds of the ipsilateral and contralateral paws, as shown in Figure 1. Among the 30 rats that received two acidic saline injections, 79.44% (n = 30) exhibited a significant reduction in paw withdrawal thresholds to mechanical stimulation with von Frey hairs 24 hours after the second injection. The remaining 20.66% (n = 6) displayed reflex responses to mechanical stimuli comparable to their pre-injection thresholds and were excluded from further analysis. On the 14th day, responders had a mean mechanical withdrawal threshold of 3.44 ± 0.012 g for the ipsilateral paw, compared to 16.55 ± 0.88 g for non-responders. Similarly, the mean withdrawal threshold for the contralateral paw was 0.97 ± 0.05 g for responders and 14.11 ± 0.1 g for non-responders. Administration of pentazocine significantly increased paw withdrawal thresholds to 10.09 ± 0.4 g for both paws in an acute study. Chronic treatment further elevated the mechanical threshold for the contralateral paw to 16.5 ± 0.3 g.
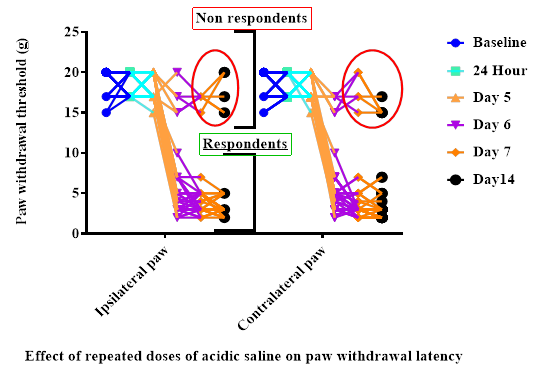
Figure 1.Effect of repeated doses of acidic saline on mechanical paw withdrawal latency. The post effect of repeated injections of acidic saline in the gastrocnemius muscle of rats (n = 36). Withdrawal threshold (g) after stimulation of the plantar surface of the hindpaw was measured with von Frey hairs and the development of mechanical hyperalgesia was observed for both the ipsilateral and contralateral hindpaws. The various time points are: baseline measured before first injection, 24 h after the first injection, immediately before second injection (on day– 5) and on day 6, 7 and 14. Data are presented as mean ± S.E.M. Number of responders was n = 31 and non-responders n = 5, to mechanical stimuli after14th day of 1st injection of acidic saline
Effect of Artemisinin on acute mechanical hyperalgesia
All tested doses of Artemisinin (5, 10, and 20 mg/kg) significantly reduced mechanical allodynia induced by acidic saline injections. In the acute study, the mechanical withdrawal threshold for the ipsilateral paw increased to 13.97 ± 0.98 g, 13.11 ± 0.90 g, and 11.98 ± 0.70 g for 5, 10, and 20 mg/kg doses, respectively (Figure 2A). For the contralateral paw, thresholds rose to 13.62 ± 0.84 g, 11.97 ± 0.70 g, and 11.75 ± 0.5 g for the same doses (Figure 2B). Pronounced effects were observed 90 minutes post-administration, with increased withdrawal thresholds for mechanical stimuli.
Effect of Artemisinin on chronic mechanical hyperalgesia
Twice-daily administration of Artemisinin for four consecutive days further reduced mechanical allodynia sensitivity. Maximum thresholds were 14.22 ± 0.4 g, 15.10 ± 0.5 g, and 15.3 ± 0.66 g for the ipsilateral paw and 16.70 ± 0.66 g, 17.30 ± 0.65 g, and 16.10 ± 0.11 g for the contralateral paw for the 5, 10, and 20 mg/kg doses, respectively (Figures 3A and 3B).
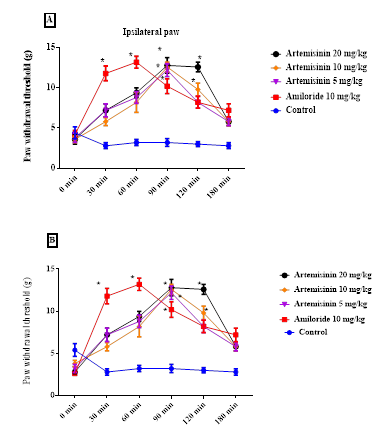
Figure 2. Effect of Artemisinin on mechanical withdrawal threshold in acute responsive studies. Artemisinin was injected immediately after the baseline responses had been obtained. Artemisinin induced an increase in ipsilateral (A) and contralateral (B) paw withdrawal threshold, which remained significantly different from baseline and vehicle treated control for up to 180 min after injection of Artemisinin and Amiloride. Data are presented as mean ± SEM, *P < 0.05 vs. corresponding vehicle time points (n=6 for each group).
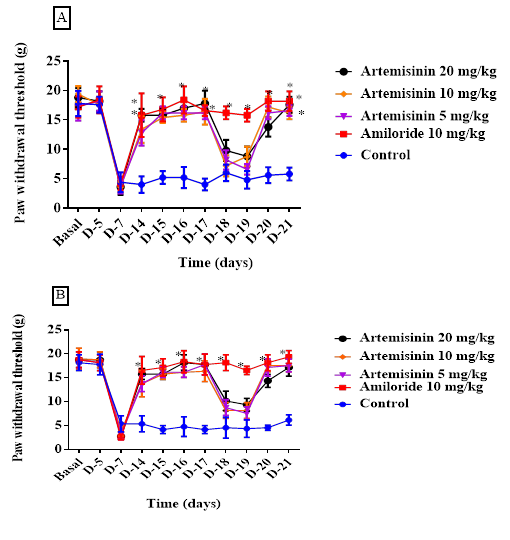
Figure 3. Effect of Artemisinin on mechanical withdrawal threshold on chronic muscle allodynia. Artemisinin induced an increase in ipsilateral(A) and contralateral (B) paw withdrawal threshold, which remained significantly different from baseline and vehicle treated control for up to 180 min after injection of Artemisinin. Data are presented as mean ± SEM,*P < 0.05 vs. corresponding vehicle time points. n=6 for each group.
Artemisinin maximum percentage effectivenesson mechanical hyperalgesia
In the acute study, Artemisinin at 20 mg/kg showed maximum percentage effectiveness (MPE) of 254.11% and 2857.0% for the ipsilateral and contralateral paws, respectively (Figure 4A). At 10 mg/kg, MPE values were 115.01% for the ipsilateral paw and 171.05% for the contralateral paw. At 5 mg/kg, MPE values were 97.00% and 110.21% for the ipsilateral and contralateral paws, respectively (Figure 4B). In the chronic study, Artemisinin at 20 mg/kg resulted in MPE values of 304.20% and 324.66% for the ipsilateral and contralateral paws, respectively (Figure 4B). At 10 mg/kg, MPE values were 209.87% for the ipsilateral paw and 331.24% for the contralateral paw, while at 5 mg/kg, MPE values were 233.11% and 238.55%, respectively.
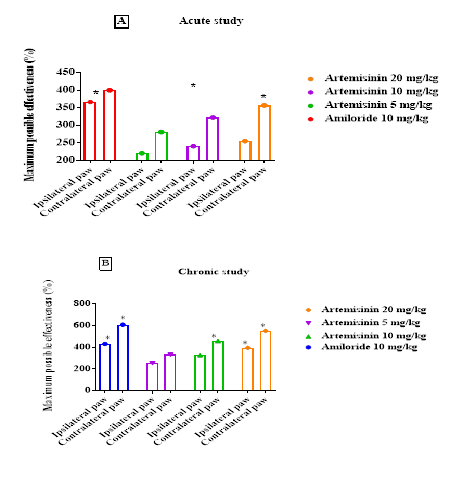
Figure 4. Maximum possible effectiveness in percentage calculated for ipsilateral and contralateral paws for the (A) Acute study and for (B) Chronic study. [The MPE values are represented only for the highest achieved values in a comparative manner; *P < 0.05 as compared to control. n=6 for each group].
Artemisinin maximum percentage effectivenesson mechanical hyperalgesia
The maximum percentage inhibition (MPI) of allodynia was 42.11%, 44.66%, and 55.22% for the ipsilateral paw and 45.10%, 48.00%, and 56.14 % for the contralateral paw, for the 5, 10, and 20 mg/kg doses, respectively (Figures 5A and 5B).The MPI of allodynia for 5, 10, and 20 mg/kg doses reached 69.02%, 77.11%, and 78.33% for the ipsilateral paw and 79.22%, 73.21%, and 77.24% for the contralateral paw (Figure 5B). Whentreatment was paused for two days (days 18 and 19), mechanical allodynia reemerged. However, resuming Artemisinin treatment on day 20 effectively reduced mechanical allodynia, suggesting no development of tolerance to the treatment.
In silico studies
Molecular Docking simulations
Based on the background literature supporting Amiloride-blockable ASICs, we conducted a theoretical analysis using molecular docking simulations. We performed molecular docking of the drug Amiloride against the target ‘an ASIC in a resting state with calcium’ (PDB ID: 5WKV) initially at pH. 7.4 (with default settings) followed by experimental conditions (in-vivo) with respect to the pH of acidic saline injection, i.e., at. pH 4.0.17additionally, to identify a potential target for ‘artemisinin’, we subjected it to docking as well. Docking results at pH. 7.4 indicated that molecule artemisinin’ (docking score: −5.814 Kcal/mol) exhibited a similar binding affinity to the drug Amiloride (docking score: −5.055 Kcal/mol).
Direct Interaction between Artemisinin and ASIC3
The drug artemisinin formed interactions with amino acid residues, such as GLU A: 374, SER A: 275, GLN A: 277, through conventional H-bonding and alkyl, π-alkyl interactions (Fig. 6).Standard Amiloride formed interactions with amino acid residues, such as GLY A: 372, LYS A:373 GLU A:374, GLU B:374, LYSB:373, GLYB: 372, SER A:275, CYS A:276, GLN A:277, CA A:504through conventional H-bonding and alkyl, π-alkyl interactions(Fig. 7). Based on the docking affinities, we conclude that Artemisinin shows a similar affinity towards ASICs as Amiloride, as demonstrated by in-silico studies.
Discussion
Fibromyalgia is a debilitating chronic pain condition with limited therapeutic options, highlighting the need for novel treatment approaches. In this study, we investigated the modulatory role of artemisinin on ASIC3 in a rat model of fibromyalgia using both in vivo and in silico approaches. Our findings demonstrate that artemisinin alleviates fibromyalgia-related pain behaviors, reduces ASIC3 expression in DRG, inhibits ASIC3 currents, and directly interacts with ASIC3 through specific binding sites in the extracellular domain.
The behavioral manifestations are consistent with previous reports [8] and reflect the ASIC3 has emerged as a promising target for pain modulation due to its involvement in various pain conditions, including inflammatory, neuropathic, and musculoskeletal pain. In line with previous studies [3-6], we observed upregulation of ASIC3 expression in DRG of fibromyalgia-model rats, supporting its role in fibromyalgia pathophysiology. Artemisinin treatment significantly reduced ASIC3 expression at both protein and mRNA levels, suggesting that downregulation of ASIC3 may contribute to its analgesic effect. Our findings provide direct evidence for artemisinin's modulatory effect on ASIC3 function. Artemisinin dose-dependently inhibited acid-evoked responses. This inhibitory effect was comparable to that of Amiloride, a selective ASIC inhibitor, suggesting that both compounds target the same channel. Additionally, artemisinin slowed the activation kinetics of acid-evoked currents without affecting desensitization kinetics, indicating that it may preferentially modulate the channel's activation process. In silico molecular docking and molecular dynamics simulation provided insights into the molecular basis of artemisinin's modulatory effect on ASIC3. The predicted binding site is located at the interface between adjacent subunits of the trimeric channel, partially overlapping with the proton-sensing region. This binding mode suggests that artemisinin may modulate ASIC3 function by interfering with proton-dependent activation, consistent with our electrophysiological observations. The stability of the artemisinin-ASIC3 complex throughout the simulation further supports the validity of the predicted binding mode.
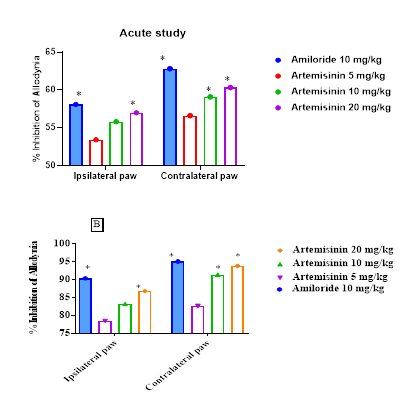
Figure 5.The percentage inhibition of Allodynia was calculated for both the ipsilateral and contralateral paws for the (A) Acute study and for (B) Chronic study. [The values are represented only for the values were the highest percentage inhibition was observed in a comparative manner; *P < 0.05 as compared to control. n=6 for each group].
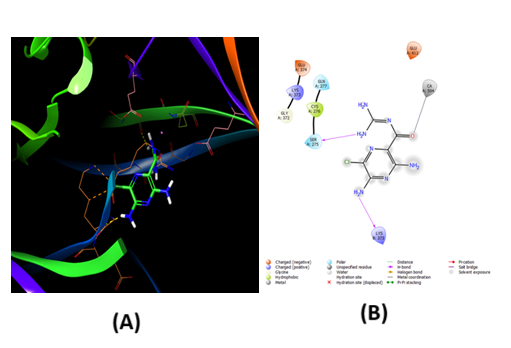
Figure. 6. (Color Online) 2D& 3D -interaction diagram of Amiloride within the binding pocket of ASIC
Table 01. Molecular Docking Results of Amiloride and Artemisinin against ASIC (PDB ID: 5WKV)
| Ligand | Docking Score (kcal/mol) | Key Interacting Residues | Interaction Types |
|---|---|---|---|
| Amiloride | −5.055 | GLY A:372, LYS A:373, GLU A:374, GLU B:374, LYS B:373, GLY B:372, SER A:275, CYS A:276, GLN A:277, CA A:504 | Conventional H-bonds, alkyl, π-alkyl interactions |
| Artemisinin | −5.814 | GLU A:374, SER A:275, GLN A:277 | Conventional H-bonds, alkyl, π-alkyl interactions |
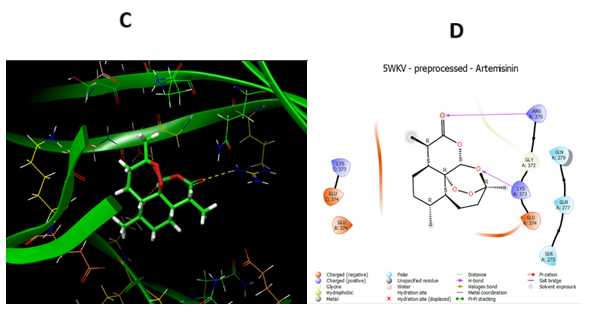
Figure 7. (Color Online) A 2D & 3D -interaction diagram of Artemisinin within the binding pocket of ASIC
Several natural compounds have been reported to modulate ASICs, including traditional Chinese medicines [14]. However, to our knowledge, this is the first study to investigate the modulatory effect of artemisinin on ASIC3 and its therapeutic potential for fibromyalgia. Our findings add to the growing body of evidence supporting the pleiotropic effects of artemisinin beyond its well-established antimalarial activity. While our results are promising, several limitations should be acknowledged. First, although we focused on ASIC3 as a potential target of artemisinin, we cannot exclude the possibility that artemisinin may exert its analgesic effect through additional mechanisms, such as anti-inflammatory actions or modulation of other ion channels involved in pain signaling. Second, we used fefemale rats in this study to reflect the higher prevalence of fibromyalgia in women; however, future studies should investigate potential sex differences in the therapeutic response to artemisinin. Third, our in silico predictions, although supported by require further validation through site-directed mutagenesis and structural studies.
Conclusion
In summary, our study demonstrates that artemisinin alleviates fibromyalgia-related pain behaviors in rats through modulation of ASIC3. This modulatory effect involves both downregulation of ASIC3 expression and direct inhibition of ASIC3 currents through specific binding to the extracellular domain of the channel. These findings suggest that artemisinin may represent a novel therapeutic approach for fibromyalgia and provide a foundation for future development of ASIC3-targeted treatments.
Materials and Methods
Animals Used: Adult Wistar female rats weighing [range 190–310 g] was used in present study. The standard environmental conditions were maintained for rats with standard diet and water ad libitum. Food and water being available freely during the experiment protocol. A prior approval protocol number RCP/P-04/24-25 was taken from by institutional animal ethical committee constituted for the purpose of control and supervision of experimental animals by ministry of Environmental and Forests, Government of India, New Delhi.
2.2. Chemical Agents- The Artemisinin (3R,5aS,6R,8aS,9R,12S,12aR)-Octahydro-3,6,9-trimethyl-3,12-epoxy-12H-pyrano[4,3-j]-1, 2-benzodioxepin-10(3H)-one was purchased (Product code: 63968-64-9) from Yucca YUCCA ENTERPRISES, Mumbai. Purity of Artemisinin was determined by the manufacturer by HPLC was certified above 98.0%. Dimethylsulphoxide (DMSO), sodium chloride (NaCl), all from Loba chemicals) were used in this investigation.
2.3. Experimental details [8]
Chronic muscular allodynia induction.- The previously described method of induction of chronic muscle-mediated pain was used. Lateral Left gastrocnemius muscle was being injected 100 microlitre of hydrochloric acid in saline solution (9 g NaCl/litre; pH adjusted to 4.0 ± 0.1). After five days the same Left lateral gastrocnemius muscle was reinjected with same protocol.
Experimental design- Female Wistar rats being grouped in five groups (6 rats per group): control, acidic saline, acidic saline with low-dose Artemisinin (5 mg/kg), acidic saline with middle dose Artemisinin (10 mg/kg) and acidic saline with high-dose Artemisinin (20 mg/kg). Mechanical allodynia, thermal allodynia, and muscle nociception were assessed at baseline and on subsequent days until end of study using behavioral tests.
The responder’s rats (n=30) were separated and divided into five groups of six animals each as follows:
- Group A (n=6) served as control group [hyperalgesic rats] injected with vehicle DMSO 0.2 ml intraperitoneally;
- Group B (n=6) served for standard drug comparison injected intraperitoneally with Amiloide at dose of 10 mg/kg,;
- Group C (n=6) served as for test drug injected intraperitoneally with Artemisinin 5 mg/kg;
- Group D (n=6) served as for test drug injected intraperitoneally with Artemisinin 10 mg/kg;
- Group E (n=6) served as for test drug injected intraperitoneally with Artemisinin 20 mg/kg;
Chronic muscle pain was induced to all the groups of animals as per the procedure described above by two injections of acidic (pH 4) saline separated by 2–5 days, given into the left gastrocnemius muscle. For evaluation of acute responses 48 hours after the second injection of acidic saline rats were treated with Amiloride, Artemisinin (5, 10 & 20 mg/kg, i.p.) or vehicle. Effects of Artemisinin was evaluated by measuring the mechanical hypersensitivity at subsequent time points. To investigate the effects of chronic treatment on mechanical hypersensitivity, Artemisinin wase administered twice a day (every 12 hours apart) to the rats for a period of 7 days. The treatment was interrupted for couple of days on the 18th and 19th day and reinitiated on 20th day to investigate the possible development of tolerance. The evaluation of nociceptive responses was performed 90 minutes after the first daily treatment (the time where the maximal inhibition was observed in the acute treatment). The animals were sacrificed on 22nd day to study the muscle histology at the site of injection.
Mechanical allodynia behavioural testing - Rats placed on elevated metal mesh grid for testing mechanical allodynia by stimulating paw plantar surface. The series of von Frey nylon hairs or filament (2–20 g) used to assess mechanical allodynia, filament applied in increasing force until the rat withdrew its paw. Each hair applied two times and the threshold (g) was noting lowest force that causes withdrawal stimuli. Prior calibrated Von Frey nylon used during entire course of the study which ensured consistency of applied forces. Rats testing mechanical withdrawal stimuli was done prior first acidic saline injection, before second injection and subsequently further on day 5, 24 hours after second injection, on day 7 for the acute study and subsequently later on from 14th day till 21st day after acidic saline second injection.
2.4. In Silico Molecular Docking
We performed molecular docking studies using the Schrödinger Suite 2023-1 (Schrödinger, LLC, New York, NY, USA), specifically using the Glide docking module through the Maestro interface.We obtained the 2D structures of Artemisinin and Amiloride from the PubChem database. These were converted into 3D structures and optimized using the LigPrep tool with the OPLS4 force field. We adjusted the pH to 4.0 using the Epik module to match the acidic environment of the biological model.We downloaded the 3D crystal structure of the acid-sensing ion channel (ASIC) from the Protein Data Bank (PDB ID: 5WKV, https://www.rcsb.org/structure/5WKV). The protein was prepared using the Protein Preparation Wizard, where we assigned bond orders, added hydrogen atoms, removed water molecules beyond 5 Å from ligands, and minimized the structure using the OPLS4 force field.We generated the receptor grid by centering it on the active site of the co-crystallized ligand with the following coordinates:X = −13.65, Y = 1.60, Z = −0.13 Å.We used the default grid box size (20 ų) to cover the binding site properly.We carried out the docking using Glide’s Extra Precision (XP) mode, which allowed for accurate sampling and scoring. We used the GlideScore (XP scoring function) to rank the ligand poses based on hydrogen bonding, hydrophobic contacts, van der Waals forces, electrostatic interactions, and desolvation penalties.To validate the docking method, we re-docked the co-crystallized ligand (2-acetamido-2-deoxy-β-D-glucopyranose) into the same binding site. The result gave an RMSD of 0.32 Å, confirming the reliability of the docking protocol.
2.5. Statistical Analysis
Statistical calculations- Graphpad Prism software version 6.01©, 1992–2012 was used for statistical calculation of obtained data. Data for mechanical allodynia are represented as mean ± SEM. One way repeated ANOVA (analysis of variance) was used to analyze the effects of Artemisinin, following Tukey’s test posthoc testing. P < 0.05 was considered to be statistically significant in all cases.
Statistical calculation for maximum possible effectiveness [%] and inhibition of allodynia [%]- The % maximum possible effectiveness and % inhibition of allodynia was calculated using equations A and B and using calculated arithmetic means in instances for detected main effect for the acute and chronic study.
a) Maximum possible effectiveness in percentage (%)-
Maximum possible effectiveness (%) = post Artemisinin treatment latency − post repeated acidic saline treatment latency × 100 (Eq. A). For present study in acute study the post latency was recorded 48 hours after second injection and for the chronic study latency was recorded 2 weeks after first injection.
Inhibitory Percentage of allodynia.
Inhibitory Percentage of allodynia (%) = test latency − mean basal withdrawal latency/15– mean basal withdrawal latency × 100 (Eq. B). In (Eq. 2) formulae the mean of the basal mechanical withdrawal threshold is calculated ipsilaterally plus contralaterally for both paws and only in responders n = 30) and the value estimated at 4.22 ± 0.70 for ipsilateral paw and 4.33 ± 0.22 for contralateral paw, to calculate % effects produced by the after intraperitoneal Artemisinin administration.
In (Eq. 2) formulae the mean of the basal mechanical withdrawal threshold is calculated ipsilaterally plus contralaterally for both paws and only in responders n = 30) and the value estimated at 4.22 ± 0.70 for ipsilateral paw and 4.33 ± 0.22 for contralateral paw, to calculate % effects produced by the after intraperitoneal Artemisinin administration.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- García-Domínguez M. Fibromyalgia and Inflammation: Unrevealing the Connection. Cells. 2025; 14(4): 271. DOI: 10.3390/cells14040271.
- Oliva V, Gregory R, Brooks JCW, Pickering AE. Central pain modulatory mechanisms of attentional analgesia are preserved in fibromyalgia. Pain. 2022; 163(1): 125–136. DOI: 10.1097/j.pain.0000000000002319
- Favretti M, Iannuccelli C, Di Franco M. Pain Biomarkers in Fibromyalgia Syndrome: Current Understanding and Future Directions. Int J Mol Sci. 2023; 24(13): 10443. DOI: 10.3390/ijms241310443
- Antonelli A, Bianchi M, Fear EJ, Giorgi L, Rossi L. Management of Fibromyalgia: Novel Nutraceutical Therapies Beyond Traditional Pharmaceuticals. Nutrients. 2025; 17(3): 530. DOI: 10.3390/nu17030530
- Siracusa R, Di Paola R, Cuzzocrea S, Impellizzeri D. Fibromyalgia: Pathogenesis, Mechanisms, Diagnosis and Treatment Options Update. Int J Mol Sci. 2021; 22(8): 3891. DOI: 10.3390/ijms22083891
- Al Sharie S, Varga SJ, Al-Husinat L, Sarzi-Puttini P, Araydah M, Bal'awi BR, Varrassi G. Unraveling the Complex Web of Fibromyalgia: A Narrative Review. Medicina (Kaunas). 2024; 60(2): 272. DOI: 10.3390/medicina60020272
- Jurado-Priego LN, Cueto-Ureña C, Ramírez-Expósito MJ, Martínez-Martos JM. Fibromyalgia: A Review of the Pathophysiological Mechanisms and Multidisciplinary Treatment Strategies. Biomedicines. 2024; 12(7): 1543. DOI: 10.3390/biomedicines12071543
- Chopade AR, Potdar VH, Mali SN, Yadav S, Pandey A, Lai CH, et al. Antifibromyalgic Activity of Phytomolecule Niranthin: In-Vivo Analysis, Molecular Docking, Dynamics and DFT. J Comput Biophys Chem. 2023; 23(3): 333–348. doi.org/10.1142/S2737416523500655.
- Li J, Zhou B. Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies. Molecules. 2010; 15(3): 1378–1397. DOI: 10.3390/molecules15031378.
- Qiao S, Zhang H, Sun F, Jiang Z. Molecular Basis of Artemisinin Derivatives Inhibition of Myeloid Differentiation Protein 2 by Combined in Silico and Experimental Study. Molecules. 2021; 26(18): 5698. DOI: 10.3390/molecules26185698
- Long Z, Xiang W, Xiao W, Min Y, Qu F, Zhang B, Zeng L. Advances in the study of artemisinin and its derivatives for the treatment of rheumatic skeletal disorders, autoimmune inflammatory diseases, and autoimmune disorders: a comprehensive review. Front Immunol. 2024; 15: 1432625. DOI: 10.3389/fimmu.2024.1432625
- Yang C, Xiong W, Dong J, Zhao X, Liang G, Zheng W. Artemisinin protected human bronchial epithelial cells from amiodarone-induced oxidative damage via 5′-AMP-activated protein kinase (AMPK) activation. Redox Rep. 2025; 30(1): 2447721. DOI: 10.3389/fimmu.2024.1432625
- Ciapała K, Rojewska E, Pawlik K, Ciechanowska A, Mika J. Analgesic Effects of Fisetin, Peimine, Astaxanthin, Artemisinin, Bardoxolone Methyl and 740 Y-P and Their Influence on Opioid Analgesia in a Mouse Model of Neuropathic Pain. Int J Mol Sci. 2023; 24(10): 9000. DOI: 10.3390/ijms241090000
- Li X, Pan H, Wang L, Zhou Q, Ma Y, Wang Q, Wang M, Xie Z, Li H, Chen L, Huang L, Chen Y, Wen C. Guidelines on Treating Fibromyalgia With Nonpharmacological Therapies in China. J Evid Based Med. 2025; 18(2): e70044. DOI: 10.1111/jebm.70044.