Summary
The progressive neurodegenerative disease known as Alzheimer's disease (AD) is characterized by tau pathology, neuroinflammation, and β-amyloid buildup. Amyloid precursor protein (APP) and presenilin-1 (PSEN1) are the main targets of classical AD research, but new study indicates that Annexin family proteins may also influence the course of the illness by regulating inflammation, membrane repair, and protein aggregation. To investigate the connection between annexins and important AD-related proteins, we used a comparative in silico technique. To find conserved areas involved in amyloidogenesis, we first performed multiple sequence alignment (MSA) of APP across Rattus norvegicus, Pan troglodytes, and Homo sapiens. Then, using MSA, BLASTp, and domain annotation tools, we examined the functional relationships and sequence similarities between Annexins (A1, A2, A4, A5, and A6) and AD-related proteins amyloid precursor protein, presenilin-1 (PSEN1) and Formyl Peptide Receptor 2 (APP, PSEN1, and FPR2). Certain annexins and AD proteins were found to be evolutionary close by phylogenetic research, indicating a potential shared ancestral origin or functional convergence. Our findings underline the possible relevance of Annexin A1, A2, and A4 as molecular contributors to disease pathophysiology and as targets for additional experimental validation by suggesting that they may be functionally and evolutionarily associated with fundamental AD pathways.
Graphical Abstract
Keywords
Alzheimer’s disease, Annexin, APP, PSEN1, FPR2, Multiple Sequence Alignment, Phylogenetics, Neurodegeneration.
Introduction
The most common type of dementia, Alzheimer's disease (AD), is typified by a buildup of abnormal protein aggregates such neurofibrillary tangles and β-amyloid plaques, as well as gradual cognitive decline and synaptic dysfunction [1](Sharmistha et al., 2025). Even though tau pathology and the amyloid cascade theory have dominated AD research for decades, new research shows that auxiliary proteins—such as the annexin protein family—that control neuroinflammatory signaling, membrane repair, vesicle trafficking, and apoptotic pathways are also significantly involved [2](Y Park et al., 2023). Recent research indicates that annexins, which are calcium-dependent phospholipid-binding proteins involved in a variety of cellular functions, may be important modulators in neurodegenerative pathways.
The ability to attach negatively charged phospholipids in a calcium-dependent way characterizes the structurally conserved family of proteins known as annexins. Each of the 12 known human annexins (ANXA1–ANXA11 and ANXA13) has a variable N-terminal region that confers functional specialization and a conserved C-terminal annexin repeat domain [3](Krzyzanowska et al., 2015). These proteins are involved in cytoskeletal architecture, endocytosis, vesicle aggregation, and membrane trafficking—processes that are becoming more and more linked to AD pathogenesis, especially in connection with synaptic degeneration, vesicle dysfunction, and membrane instability [4](Hitchcock et al., 2014).
Annexins A1, A2, A4, A5, A13 and A6 have demonstrated distinct expression patterns and regulatory roles in the pathophysiology of Alzheimer's disease. By suppressing NF-κB signaling and lowering microglial activation, for example, Annexin A1 is becoming more well acknowledged for its anti-inflammatory properties, which may provide neuroprotection in AD models [5](Han et al., 2020). On the other hand, AD patients' brains have higher levels of Annexin A5, which is known for its function in apoptotic cell clearance and may be connected to plaque buildup and neuronal death [6](Sohma, H.,et al, 2013). Notably, Annexin A4 has been implicated in amyloidogenic pathways by being connected to membrane repair and the formation of amyloid fibrils on lipid surfaces [7](Piljić & Schultz, 2006).
Annexins play a significant role in AD through their impact on amyloid formation and processing. Studies have demonstrated that in pathological settings, particularly in the lipid-rich milieu of neuronal membranes, Annexin A5 and Annexin A6 can directly bind Aβ peptides and encourage fibril formation [8](Sadleir et al, 2025). In addition to encouraging plaque nucleation, these interactions may compromise membrane integrity, resulting in calcium dyshomeostasis and neuronal damage. Furthermore, Annexin A4 seems to have two functions: it helps repair membranes under stress and, ironically, increases Aβ aggregation under chronic conditions. This could be because of oxidative stress and modifications in calcium influx [7] (Piljić & Schultz, 2006).
Chronic neuroinflammation, which is primarily caused by activated microglia and astrocytes, is another key aspect of AD pathogenesis. Annexin A1 (ANXA1) has become a crucial anti-inflammatory mediator in this regard. It limits microglial overactivation, inhibits NF-κB signaling, and facilitates the removal of debris and dead neurons by acting through the formyl peptide receptor 2 (FPR2) [9](Ansari et al., 2021). Pharmacological mimetics of ANXA1 can restore homeostasis and enhance cognitive performance in AD models, while animal studies have shown that ANXA1 deficiency worsens neuroinflammation and speeds up cognitive decline [10](Chua et al., 2022) This makes ANXA1 a promising therapeutic target in addition to being a crucial regulator of neuroimmune interaction.
Furthermore, annexin function is closely related to the disruption of calcium homeostasis, a known factor in AD. As calcium buffers or sensors, annexins—especially A2 and A6—take role in exocytosis, endocytosis, and vesicle trafficking. In AD brains, disruption of these mechanisms has been linked to axonal degeneration and decreased synaptic plasticity [11] (Torres & Cardenas, 2020). Given that synaptic loss is the most reliable indicator of cognitive impairment in AD, annexins' pathogenic significance is highlighted by their function in preserving synaptic vesicle recycling and membrane integrity.
New proteomic and transcriptome research has revealed that a number of annexins exhibit varying levels of expression in the postmortem brain tissues and cerebrospinal fluid (CSF) of AD patients, indicating their potential use as diagnostic biomarkers. For instance, elevated levels of Annexin A5 and Annexin A2 have been identified in AD CSF and have been linked to Braak stage and Aβ load [12](Weiss et al., 2016). These results pave the way for the application of annexin-based markers in progression tracking or early diagnosis [13] (de Souza Ferreira, et al, 2023).
In addition to plaque buildup, annexins also have a role in mitochondrial dysfunction, oxidative stress, poor clearance, and persistent neuroinflammation, all of which contribute to the neurodegenerative development of AD. Their significance in disease development is further expanded by their interactions with tau protein aggregation, vesicle-mediated transport regulation, and microglial phagocytic capability [14](Ries et al., 2021)
When considered collectively, the annexin family proteins are now understood to be active regulators of the various cellular and molecular processes that underpin Alzheimer's disease rather than passive structural elements. Further investigation into their various roles could provide fresh perspectives on the pathophysiology of disease and aid in the creation of annexin-targeted treatment plans [15](Jing, J. (2024)
Results
The protein and sequence retrieval of Annexin A2 (ANXA2) gave its PDB number of 1BT6 and that of Annexin A4 (ANXA4) gave its PDB number as 1ANN. The proteins shown entirely in cartoon representation, with its alpha-helices prominently displayed in green, which dominate its secondary structure as represented in figure 1. This helical architecture is characteristic of annexins, forming a conserved core that facilitates calcium-dependent binding to phospholipid membranes. The absence of beta-sheets aligns with known structural profiles of both ANXA2 and ANXA4. The positively charged red markers indicate potential calcium-binding or structurally relevant regions contributing to membrane interaction or protein–protein interaction.
Annexin A2 (ANXA2) and Annexin A4 (ANXA4) are calcium-dependent phospholipid-binding proteins involved in membrane dynamics, vesicle trafficking, and cellular signaling. Their functional association with key neurodegeneration-related proteins APP, FPR2, and PSEN1 suggests potential regulatory roles in neuroinflammatory and amyloidogenic pathways. ANXA2 has been reported to influence APP processing and trafficking, possibly modulating amyloid-beta generation, while also participating in actin remodeling and endocytosis—processes disrupted in Alzheimer’s disease. ANXA4, known for its membrane-stabilizing role, may contribute to cellular defense against oxidative and excitotoxic stress seen in neurodegeneration. Both annexins also intersect with inflammatory signaling: ANXA2 can regulate pathways linked to FPR2, a receptor involved in resolution of inflammation, while altered annexin expression may affect or reflect changes in PSEN1 function, a core component of the γ-secretase complexes that processes APP. Together, these interactions suggest that ANXA2 and ANXA4 could modulate or respond to amyloidogenic and inflammatory stress, linking membrane homeostasis to Alzheimer’s-related signaling networks.
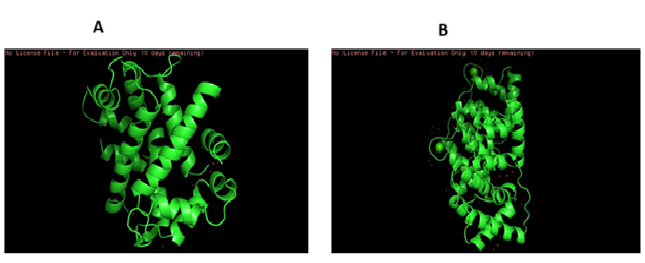
Figure 1. Three-dimensional conformation XRD structure of a) human Annexin A2 (ANXA2) and human Annexin A4 (ANXA4) visualized using PyMOL
The Multiple Sequence Alignment studies of ANXA4 across species like rat, human and chimpanzee as shown in Figure 2 help us to understand high conservation across the sequences which reflect the evolutionary conservation of structural and functional domains in Annexin A4. The multiple sequence alignment presented corresponds to Annexin A4 (A4) from rat, human, and chimpanzee, as indicated by the sequence identifiers (A4_RAT, A4_HUMAN, A4_PANTR). The alignment reveals a high degree of sequence conservation across all three species, particularly in functional domains. Notably, residues from positions 121 to 180 are perfectly conserved, indicating a functionally important domain, likely contributing to calcium-dependent membrane binding or interaction with other proteins. These conserved motifs may reflect structural domains such as annexin repeats that are central to annexin function. Additional stretches, especially toward the C-terminal end (e.g., 180–240), also exhibit strong conservation with only minor substitutions, suggesting evolutionary pressure to maintain structural integrity and function. The high conservation across mammals emphasizes the crucial, possibly non-redundant, cellular roles of Annexin A4 in membrane-related processes such as exocytosis, inflammation, or calcium signaling.
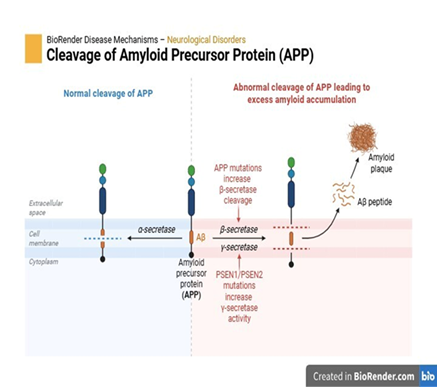
Multiple sequence alignment of the C-terminal region (residues 580–770) of APP (Amyloid Precursor Protein) from human, chimpanzee, and rat is represented in Figure 3. The alignment highlights the conserved Aβ region (residues ~672–713, shaded red) and the transmembrane domain (residues ~700–723, shaded purple). High conservation is observed across species, especially within the Aβ and transmembrane regions, emphasizing their crucial role in amyloidogenic processing and membrane insertion. Minor variations are present in the flanking sequences, while the core pathogenic region of Aβ remains highly conserved, underscoring its functional and pathological relevance across mammals.
The PPI (Protein-Protein Interaction) of the Annexin family proteins and Alzheimer’s Disease related proteins like PSEN1, FPR2, APP show a strong correlation with ANXA1, ANXA4, ANXA2, ANXA5, ANXA6, ANXA13 as shown in Figure 4. This central positioning of ANXA2 suggests it plays a pivotal role in bridging annexin-regulated processes like vesicle trafficking, membrane repair, and calcium signaling with AD-related pathways such as amyloid precursor protein processing and γ-secretase activity. ANXA1 and ANXA6 also show direct links to APP, implicating them in modulating APP cleavage and neuroinflammatory responses. ANXA5, known for its role in membrane dynamics and apoptosis, and ANXA13, a less characterized but evolutionarily conserved annexin, interact within this network—likely contributing to structural and regulatory support. The interaction of APP and ANXA1 with FPR2 further highlights annexins' involvement in inflammation-related signaling. Overall, this network underscores the functional convergence of annexins on central AD mechanisms, particularly through ANXA2, and suggests that even underexplored members like ANXA13 may play supporting roles in neurodegenerative pathways.
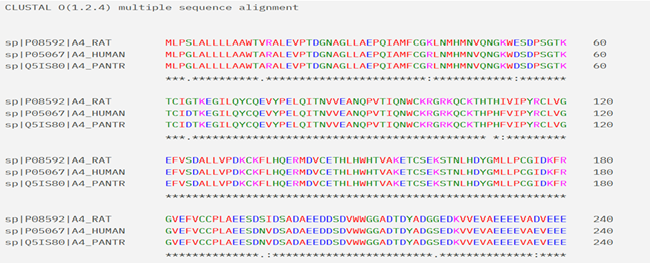


Figure 2. Multiple sequence alignment of Annexin A4 from rat, human, and chimpanzee using Clustal Omega (version 1.2.4). The sequences of Annexin A4 from Rattus norvegicus (A4_RAT), Homo sapiens (A4_HUMAN), and Pan troglodytes (A4_PANTR) were aligned to identify conserved and variable regions. Asterisks (*) indicate positions with fully conserved residues across all three species, colons (:) denote conservation between groups with strongly similar properties, and periods (.) mark weaker similarities. High conservation across the sequences reflects evolutionary conservation of structural and functional domains in Annexin A4.

Figure 3. This figure shows a multiple sequence alignment of the APP protein across human, chimpanzee, and rat, highlighting the conserved Aβ and transmembrane regions. Highly conserved regions, especially from residues 670 to 720, are shaded consistently in purple indicating potential functional or structural importance, suggesting evolutionary conservation with slight species-specific divergence.
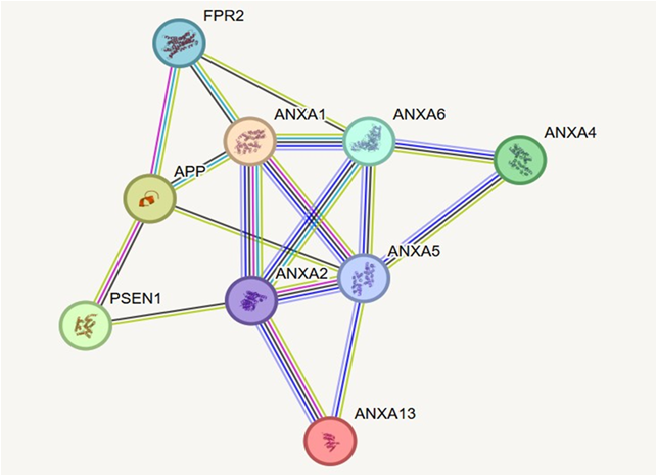
Figure 4. The protein-protein interaction (PPI) network reveals a tightly connected annexin subnetwork with ANXA2 (Annexin A2) at its core, interacting extensively with other Annexins (ANXA1, ANXA4, ANXA5, ANXA6, and ANXA13) as well as key Alzheimer’s disease (AD) proteins APP and PSEN1.
The Multiple Sequence alignment studies of Annexin family proteins (ANX) related to AD revealed a high degree of conservation across the annexin protein family, particularly in the core annexin repeat regions known to mediate calcium-dependent phospholipid binding. Conserved residues are evident across all sequences, suggesting functional importance in membrane-related processes. Variable regions, typically located in the N-terminal or loop domains, may account for isoform-specific functions and interactions with disease-associated proteins such as APP and PSEN1. ANXA2, which shows strong centrality in the PPI network, shares substantial identity with ANXA1, ANXA5, and ANXA6, supporting their overlapping roles in Alzheimer's disease pathways. ANXA13 retains many conserved motifs but also exhibits unique sequence features, reflecting its evolutionary divergence and potential for distinct cellular roles. Annexins are characterized by conserved core domains comprising four homologous repeats (I–IV), which form the structural backbone essential for their calcium-dependent membrane-binding activity. These repeats contain signature motifs such as GAGTDE, DTSGDF, and KGLGT, which are highly conserved across the annexin family, highlighting their critical functional roles. In contrast, the N- and C-terminal regions display considerable variability among different annexins, contributing to isoform-specific functions, localization, and regulatory mechanisms. For instance, ANXA6 uniquely possesses additional repeats (V–VIII), granting it extended functional capabilities compared to other annexins. Meanwhile, ANXA13 exhibits distinct sequence insertions and deletions, identifying it as the most evolutionarily divergent member of the family. The phylogenetic tree in Figure 5 reveals two main clades: ANXA1/ANXA2 and ANXA4/ANXA5/ANXA6, indicating close evolutionary and functional relationships within each group. ANXA13 branches separately with the highest divergence, reflecting its distinct evolutionary path and specialized roles. ANXA6 shows moderate divergence, positioning it between the two clusters as a possible evolutionary intermediate.
Phylogram (ancestral relationship) studies of the Annexin family proteins involved in AD
The annexin family members group into distinct clades, as shown Figure 5 with ANXA1 and ANXA2 and ANXA4 and ANXA5 showing the highest similarity
within their respective pairs, while ANXA13 is the most evolutionarily distinct. This suggests functional conservation within clades and
potential specialization in ANXA13
Analysis of functional relationship using PROSITE
Prosite analysis of individual Annexin members helps identify conserved domains, functional motifs, and signature sequences unique or shared
among the family. This allows you to understand differences in calcium-binding sites, membrane-binding regions, and isoform-specific
functional elements, ultimately revealing how each Annexin may contribute differently to cellular processes such as signaling, vesicle
trafficking, or stress response.
Prosite analysis of ANXA1 revealed four conserved regions matching known annexin domains, all aligned with its functional role in calcium-dependent membrane binding. The strongest match was observed between positions 114–185 (score: 31.258), indicating a highly conserved annexin repeat likely involved in calcium binding or structural stabilization. Additional conserved segments at positions 42–113 and 273–344 also suggest domains critical for phospholipid interaction and regulatory functions. A moderately conserved region from 197–269 may contribute to protein–protein interactions or structural integrity. Together, these matched regions confirm ANXA1’s role in membrane dynamics and signalling through well-preserved annexin domains.
Prosite analysis of ANXA2 reveals four key segments, each contributing uniquely to its structure and function. The region spanning residues 105–176 shows the highest conservation (score: 32.899) and corresponds to the core annexin domain essential for calcium-dependent phospholipid binding, underscoring its central role in membrane dynamics and signaling. The N-terminal region (33–104) is enriched in charged residues and likely aids in initial membrane association. In contrast, the segment from 189–261, with a lower score, may represent a flexible or regulatory region involved in protein-protein interactions or post-translational modifications. The final segment (265–336) contributes to oligomerization and cytoskeletal interactions. Together, these domains reflect ANXA2’s functional versatility in processes such as endocytosis, exocytosis, inflammation, and potentially neurodegenerative diseases like Alzheimer’s.
Prosite analysis of the ANXA4 protein reveals four key conserved domains contributing to its structural and functional integrity. The region from residues 14–85 (score: 28.874) appears moderately conserved and may be involved in early folding or membrane association. The segment from 86–157 shows a slightly higher score (29.548), likely corresponding to part of the annexin core domain important for calcium and phospholipid binding. Residues 169–241 (score: 28.567) maintain structural continuity within the core repeats, supporting calcium-dependent membrane interactions. The most conserved region, spanning residues 245–316 with the highest score (30.806), likely includes a critical portion of the C-terminal domain responsible for membrane affinity and potential interaction with other cellular components. Together, these conserved motifs suggest a consistent annexin repeat architecture essential for ANXA4’s membrane-binding and regulatory functions.
Prosite analysis of ANXA5 highlights four conserved segments critical to its structure and Alzheimer’s disease (AD)-related functions. The highest-scoring region, residues 15–86 (score: 30.767), encompasses the annexin core domain essential for calcium-dependent phospholipid binding and membrane association, aligning with ANXA5’s role in apoptosis and vesicle trafficking. The adjacent segment, 87–158 (score: 29.249), also shows strong conservation, rich in charged residues that likely facilitate Ca²⁺ coordination and early apoptotic recognition—key during neuronal stress in AD. The moderately conserved region from 170–242 (score: 25.393) may support protein-protein interactions, possibly with Aβ or cytoskeletal partners, while the C-terminal segment (246–317; score: 27.747) likely contributes to membrane stabilization or vesicle fusion. These conserved domains collectively reflect ANXA5’s involvement in apoptosis, Aβ interaction, membrane repair, and neuroinflammation—processes central to AD pathogenesis.
Prosite-based domain analysis of ANXA6 reveals a structurally and functionally diverse protein with eight conserved segments supporting its unique dual-core architecture. The N-terminal segments (residues 20–163; scores ~29.6) form the first annexin core, enriched in calcium-binding motifs critical for membrane association and curvature sensing—vital for neuronal vesicle trafficking. The central regions (175–322) show moderate conservation, likely mediating protein interactions or isoform-specific regulation. A strikingly conserved second core spans residues 363–506, with scores >31, highlighting its essential role in membrane fusion, calcium buffering, and neuroprotection. The less conserved region (521–595; score: 22.533) may serve regulatory functions, while the C-terminal segment (599–670; score: 30.53) reinforces membrane stabilization and cytoskeletal anchoring. These features support ANXA6’s multifaceted roles in neurophysiology and its potential contribution to Alzheimer’s disease mechanisms.
Although ANXA13 lacks direct associations with Alzheimer’s disease (AD), its evolutionary ancestry and conserved core domains suggest potential indirect roles. It may act as a functional surrogate for annexins like ANXA2 or ANXA5 under cellular stress, contributing to membrane repair, endocytosis/exocytosis, or calcium regulation—processes disrupted in AD. Additionally, its involvement in lipid raft dynamics and compensatory expression during neuroinflammation could influence neuronal stability and survival in disease contexts.
Each ribbon diagram in Figure 6 illustrates the conserved α-helical architecture characteristic of annexins, with ANXA6 and ANXA13 displaying extended structures due to additional or divergent repeats. ANXA6 shows two tandem core domains, while ANXA13 reveals structural features indicative of evolutionary divergence. These models highlight both structural conservation across annexins and isoform-specific adaptations relevant to their cellular functions, including membrane interaction and signaling in neurodegenerative contexts such as Alzheimer’s disease.
Limitations
These findings are purely based on computational results and in-silico studies which require validation through applied research. In-silico studies offer valuable support for drug discovery, toxicological assessment, prediction of physicochemical properties of proteins and ligands, it thus enhances our prediction score of the biological activities of genes, proteins and their functional dependencies. However, these predictions cannot completely be relied on keeping in mind the complexity of physiological mechanisms of biological systems. Thus, integrated workflows of using in-silico approaches together with wet lab strategies like genetic knockdown and chemical knockdown approaches on cell lines and animal models enhance efficient and effective target identification using minimal time, effort and resources. Web based systems integrating literature mining across authentic published work and curated databases accelerate the process of hypothesis testing on gene-protein relation and function, which needs an extensive. These computational tools complement wet lab experiments offering efficient means to explore and challenge complex mechanisms of biological systems.

Figure 5. Phylogenetic tree of selected human annexin (ANXA) proteins. The tree shows the evolutionary relationships among six ANXA family members: ANXA1, ANXA2, ANXA4, ANXA5, ANXA6, and ANXA13, with their corresponding UniProt accession numbers. Branch lengths are proportional to sequence divergence, and the scale bar represents a genetic distance of 0.20. The numerical values indicate the pairwise evolutionary distances for each sequence. The decimal numbers (e.g., 0.247788) are branch lengths, which indicate the evolutionary distance (typically based on % sequence divergence).
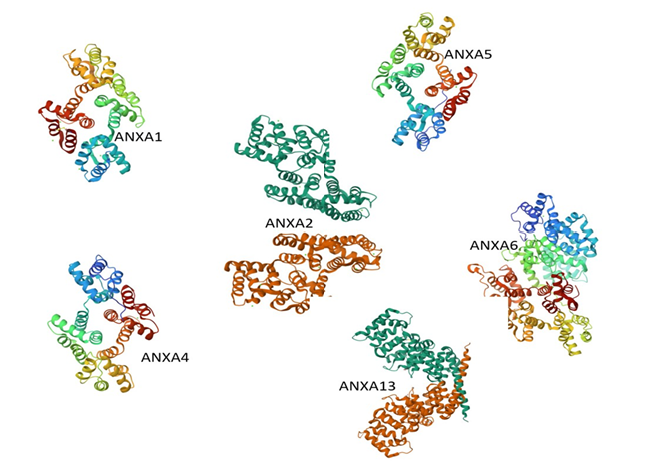
Figure 6. Comparative 3D structural representations and analysis of selected annexin family members (ANXA1, ANXA2, ANXA4, ANXA5, ANXA6, and ANXA13).
Conclusion
Previous research has mostly linked annexins to neuroinflammation and cellular balance. However, our results show that annexins and core AD proteins have always had a conversation with each other, which may be what sets off the first signs of the disease. According to the co-alignment and phylogenetic links found in this study, annexins may be upstream modulators affecting APP processing and plaque dynamics rather than only downstream effectors. Crucially, conserved domains and similar motifs suggest that annexin and AD proteins may interact directly, which calls for structural and biochemical confirmation. These revelations raise the possibility of annexin-based molecular therapies that focus on the early stages of the illness, before obvious plaque development or cognitive deterioration. Furthermore, our findings open the door for the development of isoform-specific biomarkers, which has hitherto been mainly unexplored, by identifying particular annexins (e.g., ANXA1, ANXA4, ANXA5, ANXA6) with strong evolutionary relationships to AD-related proteins. To better understand their functional significance and therapeutic potential, future studies should concentrate on experimentally analyzing these protein–protein interactions, ideally in animal and cellular models that are relevant to the disease..
Materials and Methods
1. Protein Sequence Retrieval:
The pdb & UniProtKB database (https://www.uniprot.org/) provided the FASTA formatted protein sequences of the human,(Homo sapiens)
rat (Rattus norvegicus), and chimpanzee (Pan troglodytes) amyloid precursor protein (APP), as well as the Alzheimer's disease-associated
proteins Presenilin-1 (PSEN1), Formyl Peptide Receptor 2 (FPR2), and several Annexin family proteins (Annexin A1, A2, A4, A5, A6, A13).
To guarantee accuracy and completeness, accession IDs were cross-checked for every species. PyMOL was used to visualize the three-dimensional
structure of annexin proteins, allowing detailed examination of their conserved domains and structural features.
2. Alignment of Multiple Sequences (MSA) and building of phylogenetic tree:
Multiple sequence alignment was carried out using Clustal Omega (https://www.ebi.ac.uk/Tools/msa/clustalo/) to evaluate sequence conservation
and divergence across APP across species, as well as between annexins and AD-associated proteins. To objective was to find conserved motifs,
divergent areas, and putative pathogenic functional domains, alignments were displayed. To assess the evolutionary links between the APP
proteins from various species and their resemblance to annexins and other proteins linked to Alzheimer's disease, a phylogenetic tree,
or phylogram, was created. For comparison, the Maximum Likelihood (ML) and Neighbor-Joining (NJ) approaches were used. Conserved
residues and domains were annotated using Jalview.
3. Analysis of the Functional Relationship:
Additionally, pairwise alignments were performed using BLASTp (NCBI Protein–Protein BLAST) with adjusted E-value thresholds
(< 1e-5) to assess sequence similarity and possible evolutionary conservation in order to investigate possible functional
associations between Annexin proteins and AD-related proteins (APP, PSEN1, FPR2). Additionally, Protein–protein
interaction (PPI) analysis was conducted using the STRING database ((https://string-db.org/) to identify functional
associations among key targets, including APP, PSEN1, and annexin family proteins (ANXA1, ANXA2, ANXA4, ANXA5, ANXA6,
ANXA13). To analyze structural conservation, PROSITE was used to compare all annexin family members. The analysis
revealed shared conserved domains characteristic of the annexin superfamily, supporting their functional similarity
and potential co-regulation in cellular processes.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Sharmistha D, Prabha M, Siva Kiran RR, Ashoka H. Enhanced in silico QSAR-based screening of butyrylcholinesterase inhibitors using multi-feature selection and machine learning. SAR and QSAR in Environmental Research. 2025 Feb 1;36(2):79-99. https://doi.org/10.1080/1062936X.2025.2466020
- Park LY, Cordeiro AM, Costa AC, Gattaz WF, Forlenza OV, Joaquim HP, Talib LL. Donepezil Down-Regulates Annexin A3 Protein in Alzheimer Disease Patients. Journal ISSN. 2023;2766:2276. https://www.jelsciences.com/articles/jbres1799.pdf
- Krzyzanowska A, Garcia-Consuegra I, Pascual C, Antequera D, Ferrer I, Carro E. Expression of regulatory proteins in choroid plexus changes in early stages of Alzheimer disease. Journal of Neuropathology & Experimental Neurology. 2015 Apr 1;74(4):359-69. Available from: https://doi.org/10.1097/NEN.0000000000000181
- Hitchcock JK, Katz AA, Schäfer G. Dynamic reciprocity: the role of annexin A2 in tissue integrity. Journal of cell communication and signaling. 2014 Jun;8(2):125-33. Available from: https://doi.org/10.1007/s12079-014-0231-0
- Han PF, Che XD, Li HZ, Gao YY, Wei XC, Li PC. Annexin A1 involved in the regulation of inflammation and cell signaling pathways. Chinese Journal of Traumatology. 2020 Apr 1;23(2):96-101. https://www.sciencedirect.com/science/article/pii/S1008127520300432
- Sohma H, Imai SI, Takei N, Honda H, Matsumoto K, Utsumi K, Matsuki K, Hashimoto E, Saito T, Kokai Y. Evaluation of annexin A5 as a biomarker for Alzheimer's disease and dementia with lewy bodies. Frontiers in aging neuroscience. 2013 Apr 5;5:15. https://www.frontiersin.org/journals/agingneuroscience/articles/10.3389/fnagi.2013.00015/full
- Piljić A, Schultz C. Annexin A4 self-association modulates general membrane protein mobility in living cells. Molecular biology of the cell. 2006 Jul;17(7):3318-28. https://www.molbiolcell.org/doi/full/10.1091/mbc.e06-01-0041
- Sadleir KR, Gomez KP, Edwards AE, Patel AJ, Ley ML, Khatri AW, Guo J, Mahesh S, Watkins EA, Popovic J, Karunakaran DK. Annexin A6 membrane repair protein protects against amyloid-induced dystrophic neurites and tau phosphorylation in Alzheimer’s disease model mice. Acta Neuropathologica. 2025 May 24;149(1):51: https://link.springer.com/article/10.1007/s00401-025-02888-1
- Ansari J, Senchenkova EY, Vital SA, Al-Yafeai Z, Kaur G, Sparkenbaugh EM, Orr AW, Pawlinski R, Hebbel RP, Granger DN, Kubes P. Targeting the AnxA1/Fpr2/ALX pathway regulates neutrophil function, promoting thromboinflammation resolution in sickle cell disease. Blood, The Journal of the American Society of Hematology. 2021 Mar 18;137(11):1538-49. https://doi.org/10.1182/blood.2020009166
- Chua XY, Chong JR, Cheng AL, Lee JH, Ballard C, Aarsland D, Francis PT, Lai MK. Elevation of inactive cleaved annexin A1 in the neocortex is associated with amyloid, inflammatory and apoptotic markers in neurodegenerative dementias. Neurochemistry international. 2022 Jan 1;152:105251. Available from: https://www.sciencedirect.com/science/article/pii/S0197018621002977
- Cuestas Torres DM, Cardenas FP. Synaptic plasticity in Alzheimer’s disease and healthy aging. Reviews in the Neurosciences. 2020 Apr 28;31(3):245-68. https://doi.org/10.1515/revneuro-2019-0058
- Weiss R, Bitton A, Shimon MB, Goldman SE, Nahary L, Cooper I, Benhar I, Pick CG, Chapman J. Annexin A2, autoimmunity, anxiety and depression. Journal of Autoimmunity. 2016 Sep 1;73:92-9. https://www.sciencedirect.com/science/article/pii/S0896841116300890
- de Souza Ferreira LP, da Silva RA, Gil CD, Geisow MJ. Annexin A1, A2, A5, and A6 involvement in human pathologies. Proteins: Structure, Function, and Bioinformatics. 2023 Sep;91(9):1191-204. https://onlinelibrary.wiley.com/doi/abs/10.1002/prot.26512
- Ries M, Watts H, Mota BC, Lopez MY, Donat CK, Baxan N, Pickering JA, Chau TW, Semmler A, Gurung B, Aleksynas R. Annexin A1 restores cerebrovascular integrity concomitant with reduced amyloid-β and tau pathology. Brain. 2021 May 1;144(5):1526-41. Available from: https://doi.org/10.1093/brain/awab050
- Jing J. The relevance, predictability, and utility of annexin A5 for human physiopathology. International Journal of Molecular Sciences. 2024 Mar 1;25(5):2865. Available from: https://doi.org/10.3390/ijms25052865



