Summary
Heart failure continues to exact a substantial global burden despite modern drug regimens. A persistent problem lies in the disruption of nitric oxide–soluble guanylate cyclase–cyclic GMP signalling, which promotes endothelial dysfunction, fibrosis and diastolic impairment. Vericiguat, an oral stimulator of soluble guanylate cyclase, restores cyclic GMP production independently of nitric oxide and thereby improves myocardial and vascular performance. This review integrates mechanistic, pharmacological and clinical evidence — including the SOCRATES and VICTORIA programmes — to evaluate the drug’s place in contemporary therapy. Vericiguat provides a modest but meaningful reduction in the composite outcome of cardiovascular death or first hospitalisation for heart failure, with an excellent safety profile and compatibility with established guideline-directed therapy. Its once-daily dosing and minimal interaction potential facilitate adherence. Although the absolute survival gain remains limited, vericiguat represents a mechanistically novel and clinically practical option for high-risk HFrEF patients with recent decompensation.
Keywords
vericiguat; heart failure; soluble guanylate cyclase; cyclic GMP signalling; VICTORIA trial; pharmacotherapy
Introduction
Heart failure (HF) is a complex clinical syndrome rather than a single pathological entity and arises from diverse structural and functional abnormalities of the myocardium. Contemporary management of heart failure with reduced ejection fraction (HFrEF) relies on a combination of guideline-directed medical therapies, including β-blockers, renin–angiotensin system inhibitors, angiotensin receptor–neprilysin inhibitors, mineralocorticoid receptor antagonists, and sodium–glucose cotransporter-2 inhibitors. These therapies have substantially improved survival and reduced hospitalisation rates. Nevertheless, a considerable proportion of patients continue to experience persistent symptoms, recurrent decompensation, and frequent rehospitalisations despite optimal treatment [1–3].
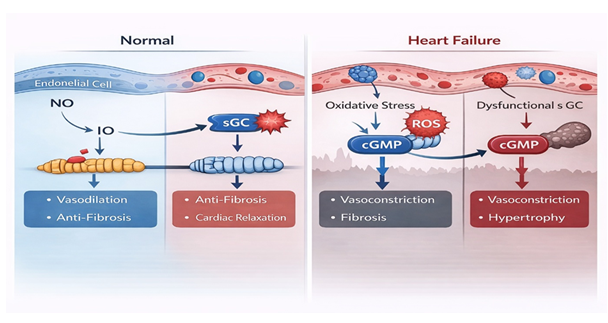
Accumulating evidence suggests that this residual risk cannot be fully explained by neurohormonal activation alone. Instead, chronic endothelial dysfunction, oxidative stress, and impaired myocardial relaxation play central roles in disease progression. Oxidative stress reduces nitric oxide (NO) bioavailability and oxidises the haem moiety of soluble guanylate cyclase (sGC), leading to diminished conversion of guanosine triphosphate to cyclic guanosine monophosphate (cGMP). Deficiency of cGMP signalling contributes to impaired myocardial relaxation, increased ventricular stiffness, progressive fibrosis, and adverse vascular remodelling [4,5].
Targeting the NO–sGC–cGMP pathway therefore represents a mechanistically distinct therapeutic strategy in heart failure. Vericiguat is an oral stimulator of soluble guanylate cyclase that directly enhances cGMP production independently of endogenous nitric oxide availability. By restoring cGMP signalling even under conditions of oxidative stress, vericiguat offers a novel approach to addressing pathophysiological processes that are inadequately corrected by conventional neurohormonal therapies [6].
This narrative review summarises the pharmacological properties of vericiguat, evaluates evidence from key preclinical and clinical studies—including the SOCRATES and VICTORIA programmes—and discusses its role in contemporary management of patients with HFrEF.
Results and Discussion
Mechanistic overview
Heart failure (HF) remains a major global health concern, characterized by high rates of morbidity, mortality, and recurrent hospitalizations, despite significant advancements in pharmacological therapies. A primary pathophysiological defect in HF is the dysfunction of the nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate (NO–sGC–cGMP) signaling pathway, which results in endothelial dysfunction, myocardial fibrosis, impaired relaxation, and persistent ventricular remodeling. Vericiguat, a novel oral stimulator of soluble guanylate cyclase, directly enhances cGMP production, independent of nitric oxide availability, thus targeting a disease mechanism that is inadequately addressed by conventional neurohormonal therapies.
This review synthesizes the current evidence regarding vericiguat, including its molecular mechanisms, pharmacokinetics, preclinical investigations, and clinical outcomes from pivotal trials, such as the SOCRATES and VICTORIA programs. Clinical results suggest that vericiguat offers a modest yet clinically meaningful reduction in the composite endpoint of cardiovascular death or the first hospitalization for heart failure in patients with worsening heart failure and reduced ejection fraction (HFrEF), along with a favorable safety and tolerability profile. Its once-daily oral dosing, minimal drug–drug interactions, and compatibility with guideline-directed medical therapy support its use in routine clinical practice. Although the absolute mortality benefits remain limited, vericiguat represents a vital mechanistically novel adjunctive option for stabilizing high-risk patients who have recently undergone decompensation.
Pharmacokinetics and pharmacodynamics
Vericiguat demonstrates advantageous pharmacokinetic characteristics that facilitate once-daily oral dosing. When ingested with
food, its oral bioavailability reaches nearly 93%, and systemic exposure shows a slight increase compared to fasting conditions.
Peak plasma levels are generally attained about one hour post-administration. The terminal elimination half-life is approximately
20 hours, allowing for stable plasma levels with daily intake. Metabolism primarily occurs through glucuronidation, facilitated by
UGT1A9 and UGT1A1, with minimal participation from cytochrome P450 enzymes. As a result, the likelihood of clinically significant
drug–drug interactions remains low. Roughly equal amounts of the administered dose are eliminated through urine and feces.
Pharmacodynamic investigations reveal a dose-dependent rise in plasma cGMP levels, indicating effective target engagement.
Notably, vericiguat induces minimal clinically significant alterations in heart rate or systemic blood pressure, setting it apart
from conventional vasodilators.

Pre-clinical and early clinical data
Preclinical studies have demonstrated that stimulation of the soluble guanylate cyclase–cGMP pathway by vericiguat leads to
favourable structural and functional cardiac effects. In experimental models of heart failure, treatment with vericiguat reduced
left ventricular hypertrophy and myocardial fibrosis while improving diastolic compliance and ventricular relaxation [18,19].
These findings provided early evidence that restoring cGMP signalling may attenuate maladaptive cardiac remodelling.
Phase I clinical studies conducted in healthy volunteers confirmed that vericiguat is well tolerated and exhibits predictable,
dose-dependent pharmacokinetics, thereby supporting further clinical development [21]. Subsequent phase II evaluation in the
SOCRATES-REDUCED trial enrolled 456 patients with HFrEF and demonstrated significant dose-related reductions in NT-proBNP
concentrations, accompanied by modest improvements in left ventricular ejection fraction [22]. In contrast, the SOCRATES-PRESERVED
trial, which included 477 patients with HFpEF, did not demonstrate significant changes in NT-proBNP levels, although some
improvements in functional measures and patient-reported outcomes were observed [23]. Collectively, these early clinical findings
highlighted the potential benefit of vericiguat in patients with reduced ejection fraction and informed the design of the pivotal
VICTORIA outcomes trial.
| Trial | Population | Design | Primary endpoint | Key result | Reference |
|---|---|---|---|---|---|
| SOCRATES-REDUCED | HFrEF (n = 456) | Randomised phase II dose-finding | Change in NT-proBNP | ↓ NT-proBNP, ↑ LVEF | [22] |
| SOCRATES-PRESERVED | HFpEF (n = 477) | Randomised phase II | Change in NT-proBNP | Neutral biomarker trend | [23] |
| VICTORIA | HFrEF (n = 5050) | Randomised phase III outcomes trial | CV death or first HF hospitalisation | HR 0.90 (95 % CI 0.82–0.98) | [8] |

The VICTORIA trial and clinical interpretation
The VICTORIA trial was a large, randomised, double-blind, placebo-controlled phase III study that enrolled 5,050 patients with
symptomatic HFrEF who had experienced recent clinical worsening, defined by hospitalisation or the need for intravenous diuretics
[8]. All participants received guideline-directed medical therapy, and vericiguat was initiated at a dose of 2.5 mg once daily,
with gradual titration to a target dose of 10 mg based on tolerability. Over a median follow-up period of 10.8 months, vericiguat
significantly reduced the primary composite endpoint of cardiovascular death or first heart-failure hospitalisation compared with
placebo (hazard ratio 0.90; 95% confidence interval 0.82–0.98; p = 0.02). The observed benefit was predominantly driven by a
reduction in heart-failure hospitalisations, whereas cardiovascular mortality alone was not significantly different between
groups. Vericiguat demonstrated a favourable safety profile, with low rates of treatment discontinuation. Mild hypotension and
anaemia were the most frequently reported adverse events, but these rarely necessitated cessation of therapy. Subgroup analyses
indicated consistent efficacy across age groups, sex, renal function categories, and background use of angiotensin
receptor–neprilysin inhibitors or SGLT2 inhibitors [24]. These findings support the role of vericiguat as a stabilising therapy
in patients with high-risk HFrEF following recent decompensation.
Comparison with established heart-failure therapies
The contemporary management of HFrEF relies on a multidrug strategy that targets complementary pathophysiological pathways.
Foundational therapies—including β-blockers, RAAS inhibitors, mineralocorticoid receptor antagonists, and SGLT2 inhibitors—have
demonstrated robust mortality and morbidity benefits across a broad range of patient populations. In contrast, vericiguat
primarily addresses impaired cGMP signalling and offers incremental benefit by reducing recurrent hospitalisations in selected
high-risk patients. Its role is therefore best understood as additive rather than foundational, complementing existing therapies
rather than replacing them.
Safety profile
The comprehensive safety profile of vericiguat has been thoroughly assessed throughout phase I to phase III clinical development
programs, as well as through post-hoc analyses of significant trials. Across various studies, vericiguat has shown good
tolerability, with adverse events typically being mild to moderate in severity and occurring at frequencies comparable to placebo
[17]. Notably, the rates of discontinuation due to adverse events were low, indicating its appropriateness for prolonged
outpatient use. The most frequently reported treatment-related adverse events consist of mild hypotension and anaemia.
Hypotension seems to be dose-dependent but is generally asymptomatic and can be managed through careful titration and adjustment
of accompanying diuretic therapy. The anaemia associated with vericiguat is believed to be a result of haemodilution rather than
bone marrow suppression or haemolysis, and significant reductions in haemoglobin levels are rare [8,17]. Serious bleeding
incidents have not been observed at rates exceeding those of placebo. Renal safety remains a critical issue for patients with
advanced heart failure, many of whom also suffer from chronic kidney disease. In the VICTORIA trial, vericiguat did not elevate
the incidence of acute kidney injury or lead to clinically significant declines in estimated glomerular filtration rate when compared to placebo, even in patients with moderate renal impairment at baseline [24]. Additionally, electrolyte imbalances, such as hyperkalaemia and hyponatraemia, were not significantly different between the treatment groups.
| Drug class | Mechanism of action | Mortality benefit | Primary clinical advantage |
|---|---|---|---|
| β-blockers | Inhibit sympathetic drive | High | Reverse remodelling and improved survival |
| ACE inhibitors/ARNI | Block RAAS and enhance natriuretic peptides | High | Improved symptoms and outcomes |
| Mineralocorticoid antagonists | Counter aldosterone-mediated fibrosis | Moderate | Anti-fibrotic effect |
| SGLT2 inhibitors | Modify metabolism and osmotic diuresis | High | Reduced admissions and mortality |
| Vericiguat | Stimulates sGC → ↑ cGMP | Modest | Fewer rehospitalisation and improved stability |

Economic and practical considerations
In addition to clinical effectiveness, the integration of any new heart failure treatment is significantly shaped by its economic
implications and practical applicability within healthcare frameworks. Heart failure ranks among the top causes of hospital
admissions globally, with repeated hospital stays constituting a considerable share of the total healthcare costs associated with
this condition. Therefore, interventions aimed at decreasing rehospitalisation rates, even in the absence of substantial
mortality improvements, may still present advantageous cost-effectiveness profiles [26]. Pharmacoeconomic evaluations performed
in Europe and North America have assessed the additional cost-effectiveness of vericiguat when used alongside guideline-directed
medical therapy. These analyses suggest an incremental cost per quality-adjusted life-year (QALY) gained ranging from £17,000 to
£20,000, which aligns with the accepted willingness-to-pay thresholds in numerous high-income healthcare systems [16,26]. Notably,
the economic value of vericiguat is primarily influenced by its ability to reduce heart failure hospitalisations rather than by
improvements in survival rates.
Practical selection criteria
Appropriate selection of patients is essential for maximizing the therapeutic advantages of vericiguat while minimizing potential
adverse effects. Findings from the VICTORIA trial and subsequent evaluations suggest that the drug's efficacy is most significant
in patients experiencing recently exacerbated heart failure, especially those who have necessitated hospitalization or intravenous
diuretic treatment within the last three months [8,24]. Haemodynamic stability is a crucial requirement for the commencement of
treatment. Patients must exhibit a systolic blood pressure of no less than 100 mmHg to mitigate the risk of symptomatic
hypotension. Vericiguat should be initiated only after the optimization of foundational guideline-directed medical therapy,
as it is designed to serve as an adjunct rather than a replacement for established medications. Elevated levels of natriuretic
peptides assist in identifying high-risk patients; however, extremely elevated NT-proBNP levels may indicate advanced disease,
where the relative benefit of vericiguat may be diminished.
| Criterion | Rationale | Suggested action |
|---|---|---|
| Recent decompensation within three months | Benefit concentrated in recently decompensated cohorts | Start once haemodynamically stable |
| Optimised guideline therapy | Vericiguat is adjunctive | Maximise tolerated GDMT before initiation |
| Systolic BP ≥ 100 mmHg | Avoid symptomatic hypotension | Delay initiation or adjust diuretics if lower |
| Baseline NT-proBNP < 8000 pg/mL | Attenuated effect at very high levels | Prioritise patients below this threshold |
| eGFR ≥ 30 mL min−1 1.73 m−2 | Limited data in severe renal impairment | Use cautiously and monitor renal function |
| No PDE-5 inhibitor use | Risk of excessive vasodilation | Contraindicated or advise against concurrent use |
Clinical vignette
The subsequent clinical case exemplifies the practical use of vericiguat in standard care. A 68-year-old male with ischaemic
cardiomyopathy and a left ventricular ejection fraction of 30% presented with exacerbating dyspnoea and peripheral oedema, which
required hospitalisation and intravenous diuretic treatment. His medical background included chronic kidney disease (estimated
glomerular filtration rate 42 mL min⁻¹ 1.73 m⁻²) and hypertension. In spite of the optimisation of guideline-directed
therapy—including an ACE inhibitor, β-blocker, mineralocorticoid receptor antagonist, and SGLT2 inhibitor—he continued to
experience symptoms at New York Heart Association (NYHA) class III. His NT-proBNP level was 4,200 pg/mL, and his blood pressure
remained stable at 110/68 mmHg. In light of his recent decompensation and ongoing risk, vericiguat was commenced at a dose of 2.5
mg once daily and was gradually increased to 10 mg over several weeks.
Author’s perspective and practical recommendations
From a clinical perspective, vericiguat should be considered a targeted therapy for patients with HFrEF who remain vulnerable to
recurrent decompensation despite optimisation of guideline-directed medical therapy. Its greatest value lies in stabilising
patients during the high-risk period following recent hospitalisation or intravenous diuretic therapy. Initiation should occur
only after haemodynamic stability is achieved, with gradual dose titration and close monitoring during early treatment.
Clinicians should set realistic expectations, emphasising that the principal benefit of vericiguat is a reduction in heart-failure hospitalisations rather than dramatic symptom improvement or survival extension. Careful patient selection, combined with structured follow-up, is essential to maximise benefit while maintaining safety. As real-world experience grows, institutional protocols and registries may further refine its optimal use.
Future directions
Current studies look to figure out vericiguat's place in heart failure where ejection fraction stays preserved, along with how
it pairs with other drugs that tweak cGMP pathways. References[27 - 30]cover those efforts. Down the line, one approach might
combine stimulation of two pathways at once through both the soluble and particulate forms of guanylate cyclase. That could add
up to better relaxation in heart muscle and more flexible blood vessels.
Limitations and Unmet Needs
Despite its mechanistically novel approach and favourable safety profile, vericiguat has several important limitations that
warrant consideration. The magnitude of benefit observed in clinical trials has been modest compared with foundational therapies
such as β-blockers, angiotensin receptor–neprilysin inhibitors, and SGLT2 inhibitors. In the VICTORIA trial, the reduction in the
composite endpoint was primarily driven by fewer heart-failure hospitalisations, while a clear mortality benefit was not
demonstrated. These findings indicate that vericiguat should be regarded as an adjunctive rather than disease-defining therapy.
Several unresolved questions remain. The optimal timing of initiation—whether during hospitalisation, immediately after discharge, or later during stable outpatient management—has not been conclusively established. Evidence supporting its use in patients with severe renal impairment, profound hypotension, or extremely elevated natriuretic peptide levels remains limited. Furthermore, access and affordability may restrict widespread use in resource-limited healthcare settings.
Future research should focus on pragmatic real-world studies, longer-term outcome analyses, and improved phenotypic patient selection to clarify the populations most likely to benefit from vericiguat. Addressing these gaps will be essential to defining its long-term role in comprehensive heart-failure management.
Mechanistic and Translational Insights
Preclinical studies suggest that stimulation of the sGC–cGMP pathway by vericiguat may exert several potentially beneficial
biological effects, including attenuation of inflammatory signalling, improvement of mitochondrial efficiency, reduction of
myocardial fibrosis, and enhancement of endothelial function. Experimental models have demonstrated downregulation of
pro-inflammatory cytokines, improved cellular energetics, and reduced fibroblast proliferation following restoration of cGMP
signalling.
However, it is important to note that most of these observations are derived from preclinical or early translational studies. Direct evidence confirming long-term disease-modifying effects in humans remains limited. Consequently, these mechanistic findings should be interpreted as hypothesis-generating rather than definitive proof of structural myocardial reversal. Further clinical studies are required to determine whether these biological effects translate into sustained improvements in cardiac structure and long-term outcomes.
Materials and methods
Search strategy
We did a planned search through PubMed, ScienceDirect, and ClinicalTrials.gov. The time frame covered 2015 to 2024. Search terms
included vericiguat. We also used sGC stimulator. Heart failure came up. SOCRATES and VICTORIA got included. Extra details came
from European Medicines Agency or EMA documents. US Food and Drug Administration or FDA files added more. References [9,10] cover
those.
Eligibility criteria
We picked studies on preclinical work with vericiguat. Phase one through three trials made the cut. Major reviews on its mechanism,
pharmacology, or outcomes worked too. We left out reports not in English. Non-peer-reviewed stuff got skipped. Studies on
unrelated drugs did not qualify.
Data synthesis
We pulled out info on study designs. Participant details came next. Interventions and endpoints followed. Safety results rounded it
out. The evidence got put together narratively. We followed the IMRaD structure from Vita Scientia publications.
Conclusion
Heart failure with reduced ejection fraction remains a progressive and burdensome clinical condition despite advances in
guideline-directed medical therapy. Although neurohormonal blockade has significantly improved outcomes, many patients continue
to experience recurrent decompensation and hospitalisation. Vericiguat represents a mechanistically distinct therapeutic option
by restoring impaired soluble guanylate cyclase–cGMP signalling, a pathway inadequately addressed by conventional therapies.
Evidence from the SOCRATES and VICTORIA programmes indicates that vericiguat provides a modest but clinically meaningful reduction in heart-failure hospitalisations, particularly among patients with recent decompensation who remain at high risk despite optimized background therapy. Its favourable safety profile, minimal drug–drug interactions, and once-daily dosing support its practical integration into clinical care.
Vericiguat should be viewed as an adjunctive therapy rather than a replacement for established treatments, with careful patient selection and realistic expectations regarding benefit. Ongoing and future studies will be essential to clarify its optimal timing, broader applicability, and potential role in other heart-failure phenotypes. When used judiciously, vericiguat adds a valuable mechanism-based option to the modern heart-failure treatment landscape.
Acknowledgement
The author expresses deep gratitude for the academic guidance, institutional support, and scholarly resources offered by Br. Harne College of Pharmacy, Thane, Maharashtra, India, which played a crucial role in the successful completion of this review. The encouragement and assistance from the faculty members of the Department of Pharmacy are genuinely appreciated, especially for promoting a research-focused academic atmosphere and providing access to pertinent scientific literature and databases. Additionally, the author acknowledges the valuable contributions of researchers and clinicians whose published works laid the groundwork for this review. Their thorough investigations and clinical insights into the pathophysiology of heart failure and the modulation of soluble guanylate cyclase have greatly enhanced understanding in this area and facilitated the synthesis presented in this document.
Author contributions
Rudra Adhik Khanolkar conceptualised the review, designed the study framework, and drafted the manuscript. Dnyaneshwar S. Vyavhare contributed to literature screening, and critical revision of the manuscript. Gita Mohire assisted in refining the clinical interpretation, reviewing pharmacological content. All authors reviewed and approved the final manuscript and accept responsibility for the integrity and accuracy of the work.
Conflict-of-interest statement
The author asserts that there are no conflicts of interest, whether financial or non-financial, associated with the research, authorship, or publication of this manuscript. The author has not obtained any honoraria, consultancy fees, speaker payments, or research funding from pharmaceutical companies engaged in the development or marketing of vericiguat or related cardiovascular therapies. All interpretations and conclusions articulated in this review are derived exclusively from an independent and critical assessment of the published scientific literature and are devoid of any commercial or institutional influence
Funding statement
This review article did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The work was conducted as part of the author’s academic activities at Br. Harne College of Pharmacy and was supported solely through institutional resources and personal scholarly effort.
Data-access statement
All data and information presented in this review were sourced from publicly available materials, such as peer-reviewed journal articles, clinical trial registries, and reports from regulatory authorities. No new datasets were created or analyzed specifically for this review. Therefore, there are no further data available for dissemination beyond the references mentioned in the manuscript.
Ethics statement
This is a narrative synthesis of previously published data and therefore, did not require ethical approval or participant consent.
References
- Paulus WJ, Tschöpe C. A novel paradigm for HFpEF. J Am Coll Cardiol. 2013;62:263–271. https://doi.org/10.1016/j.jacc.2013.02.092
- Tsutsui H et al. Oxidative stress in cardiac failure. Circ J. 2020;84:1030–1038. https://doi.org/10.1253/circj.CJ-19-0951
- Sandner P et al. Soluble guanylate cyclase stimulators and activators. Handb Exp Pharmacol. 2021;264:355–394. https://doi.org/10.1007/164_2021_389
- Gheorghiade M et al. Pharmacological modulation of cyclic GMP signalling. Eur Heart J. 2019;40:2900–2910. https://doi.org/10.1093/eurheartj/ehz273
- Armstrong PW et al. Vericiguat in patients with heart failure and reduced ejection fraction. N Engl J Med. 2020;382:1883–1893. https://doi.org/10.1056/NEJMoa1915928
- European Medicines Agency. Verquvo (vericiguat) Assessment Report. EMA; 2021.
- US Food and Drug Administration. Summary Review: Vericiguat Tablets. FDA; 2021.
- Stasch JP et al. Pharmacology of sGC modulators. Circulation. 2015;131:1074–1085. https://doi.org/10.1161/CIRCULATIONAHA.114.013540
- Münzel T et al. Pathophysiology of oxidative stress and cGMP deficiency in HF. Br J Pharmacol. 2018;175:2310–2324. https://doi.org/10.1111/bph.14200
- Evgenov OV et al. NO-independent activation of soluble guanylate cyclase. Nat Rev Drug Discov. 2011;10:755–769. https://doi.org/10.1038/nrd3562
- Bender AT, Beavo JA. cGMP signalling: physiology and pharmacology. Pharmacol Rev. 2006;58:220–264. https://doi.org/10.1124/pr.58.2.3
- Filippatos G et al. Vericiguat: a novel therapeutic option. Eur Heart J. 2022;43:451–458. https://doi.org/10.1093/eurheartj/ehab912
- Wirtz J et al. Pharmacoeconomic evaluation of vericiguat. Health Econ Rev. 2022;12:37–48. https://doi.org/10.1186/s13561-022-00370-8
- Butler J et al. Safety and tolerability of vericiguat. Clin Cardiol. 2019;42:636–643. https://doi.org/10.1002/clc.23178
- Ghofrani HA et al. Role of cGMP signalling. Br J Pharmacol. 2017;174:1771–1784. https://doi.org/10.1111/bph.13654
- Liu S et al. Vericiguat ameliorates cardiac fibrosis. Cardiovasc Ther. 2020;38:e12491. https://doi.org/10.1111/1755-5922.12491
- McMurray JJV et al. Pathophysiological targets beyond neurohormonal blockade. Nat Rev Cardiol. 2022;19:706–722. https://doi.org/10.1038/s41569-022-00691-z
- Stasch JP et al. NO-independent activation of sGC. Trends Pharmacol Sci. 2015;36:163–172. https://doi.org/10.1016/j.tips.2015.01.002
- Armstrong PW et al. Predictors of outcome in the VICTORIA trial. Eur J Heart Fail. 2022;24:186–198. https://doi.org/10.1002/ejhf.2394
- Solomon SD et al. VITALITY-HFpEF trial. Eur J Heart Fail. 2021;23:1264–1273. https://doi.org/10.1002/ejhf.2249
- Lam CSP et al. Translating cGMP biology into clinical benefit. Heart Fail Rev. 2021;26:713–724. https://doi.org/10.1007/s10741-020-09999-0
- Halliday BP et al. Future directions in heart-failure pharmacotherapy. Nat Rev Cardiol. 2024;21:201–218. https://doi.org/10.1038/s41569-023-00939-y
- Pandey A et al. Economic burden and cost-effectiveness. J Am Coll Cardiol. 2021;77:2193–2205. https://doi.org/10.1016/j.jacc.2021.02.027
- Papp Z et al. Emerging molecular targets for myocardial relaxation. Br J Pharmacol.
- 2020;177:2072–2090. https://doi.org/10.1111/bph.14960
- Boehm M et al. Dual-pathway cGMP stimulation in heart failure. Cardiovasc Drugs Ther. 2023;37:447–458. https://doi.org/10.1007/s10557-023-07329-2
- Butler J et al. Vericiguat in HFpEF. Eur Heart J Suppl. 2022;24:L33–L38. https://doi.org/10.1093/eurheartjsupp/suac053
- Konstam MA et al. VITALITY-HFpEF results. Eur Heart J. 2023;44:1192–1201. https://doi.org/10.1093/eurheartj/ehad023



